|
11/24/2023 0 Comments Understanding precipitate chemistry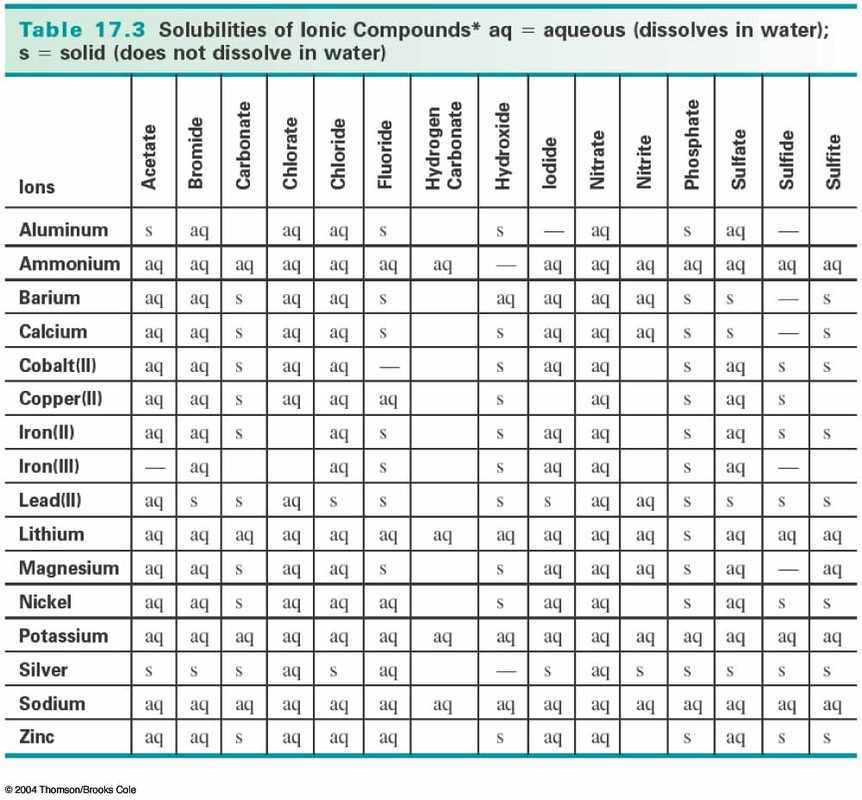
When two aqueous solutions are mixed, the ions interact to form products. Why does a precipitate form for some solutions? If some of the solid is added, the excess ions precipitate out and until solubility equilibrium is achieved. 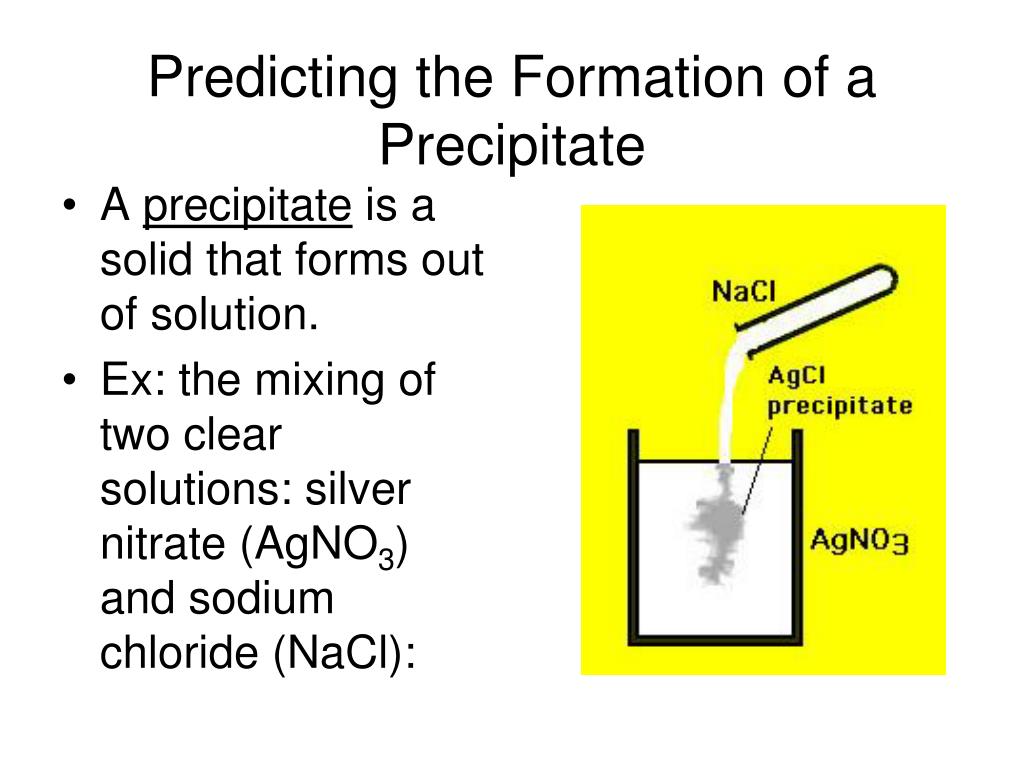
How does a precipitate affect equilibrium?Ī supersaturated solution is not at equilibrium, and no solid can ordinarily be present in such a solution. In a chemical laboratory it usually refers to a solid crystallizing from a liquid solution, but in weather reports it applies to liquid or solid water separating from supersaturated air. 
Precipitation is a process in which a solute separates from a supersaturated solution. So, when a calcium chloride (one example of Ca+2) and oxalic acid are each dissolved in water and then mixed, the reaction in Figure 1 occurs instantaneously forming the calcium oxalate which precipitates out of solution. Is a kidney stone a precipitation reaction? Chemical reactions occur everywhere in the world around you, not just in a chemistry lab. What is chemical reaction everyday life?Įxamples of chemical reactions in everyday life include photosynthesis, rust, baking, digestion, combustion, chemical batteries, fermentation, and washing with soap and water. The reaction between silver nitrate (AgNO3) and sodium chloride (NaCl), results in the formation of silver chloride (AgCl) that is an insoluble salt. Some examples are reaction between calcium chloride (CaCl2) and potassium hydroxide(KOH), resulting in the formation of calcium hydroxide that is an insoluble salt. What are two examples of precipitation reactions? Fairly uniform precipitation composed exclusively of fine drops very close together. Most commonly observed, drops larger than drizzle (0.02 inch / 0.5 mm or more) are considered rain. In this example, the precipitate is silver chloride. Precipitation Example Mixing silver nitrate and sodium chloride in water will cause silver chloride to precipitate out of solution as a solid. What is a precipitate in chemistry examples? It is highly soluble in water and has high ionic strength. Which of the following can be used for selective precipitation of proteins? Explanation: Ammonium sulfate is commonly used for selective precipitation of proteins. Which of the following can be used for selective precipitation of proteins? If a precipitate is formed when a chemical reacts with lead, for example, the presence of lead in water sources could be tested by adding the chemical and monitoring for precipitate formation. Precipitation reactions are useful in determining whether a certain element is present in a solution. Why is precipitation important in chemistry? Qualitative Analysis for Metallic Elements. Selective precipitation is a technique of separating ions in an aqueous solution by using a reagent that precipitates one or more of the ions, while leaving other ions in solution. What is selective precipitation in chemistry? In wastewater, a frequent presence of heavy metals can be found such as compounds of sulphide and hydroxide. 
When a contaminant forms an insoluble solid, then we can use this reaction to precipitate out the contaminated ions. Precipitation reaction can be used in wastewater treatment. How precipitation reactions are used in the real world? Selective precipitation of proteins can be used as a bulk method to recover the majority of proteins from a crude lysate, as a selective method to fractionate a subset of proteins from a protein solution, or as a very specific method to recover a single protein of interest from a purification step. 24 How do kidney stones form chemistry? What is selective precipitation used for?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
